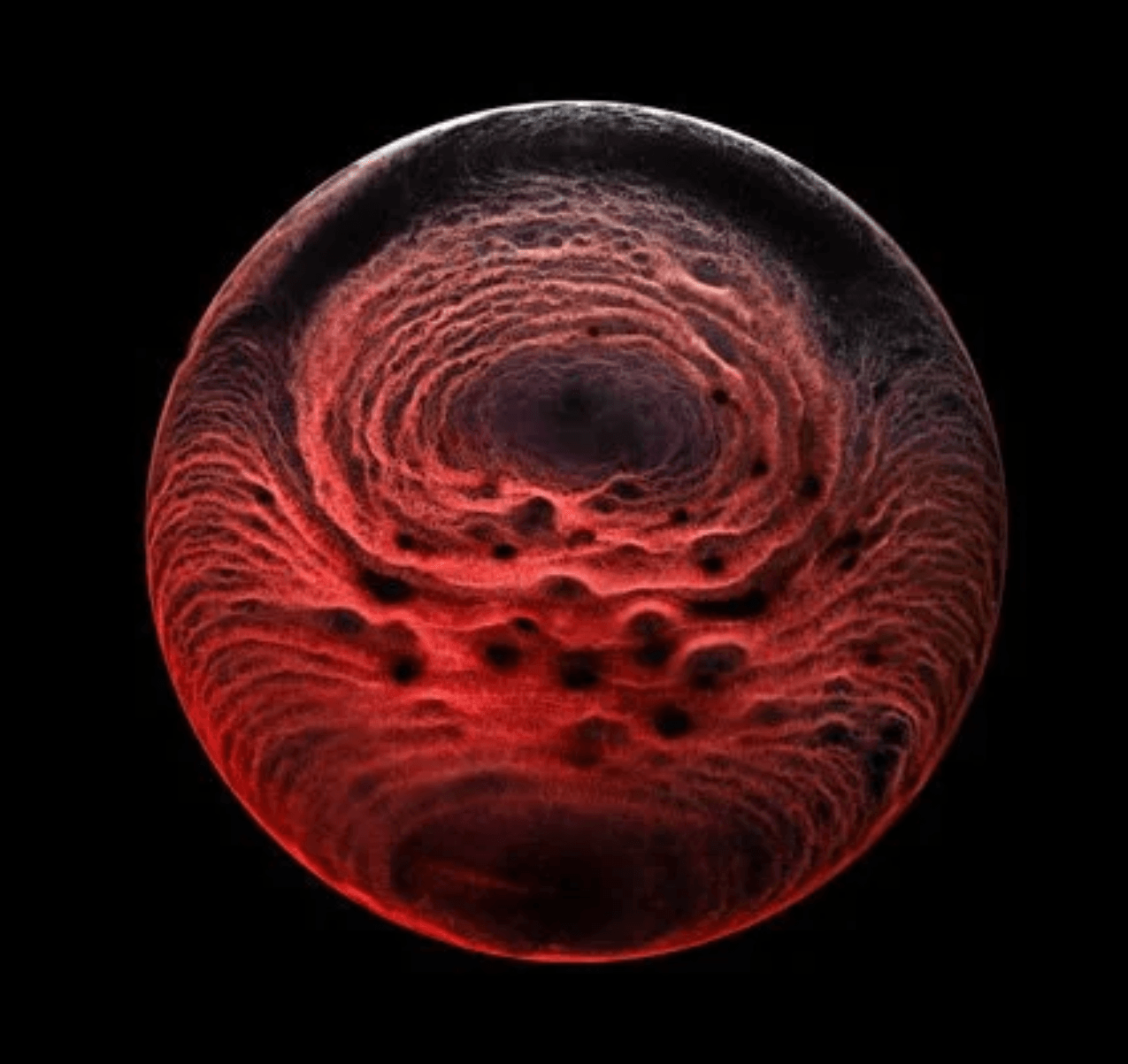
Photographer Ernie Button explores the stains left behind when various liquors evaporate. This one comes from a single malt scotch whisky by The Balvenie. The stain itself is made up of particles left behind when the alcohol and water in the whisky evaporate. The pattern itself depends on a careful interplay between surface tension, evaporation, pinning forces, and internal convection as the whisky puddle dries out. (Image credit: E. Button/CUPOTY; via Colossal)
Tag: alcohol

Whiskey Webs
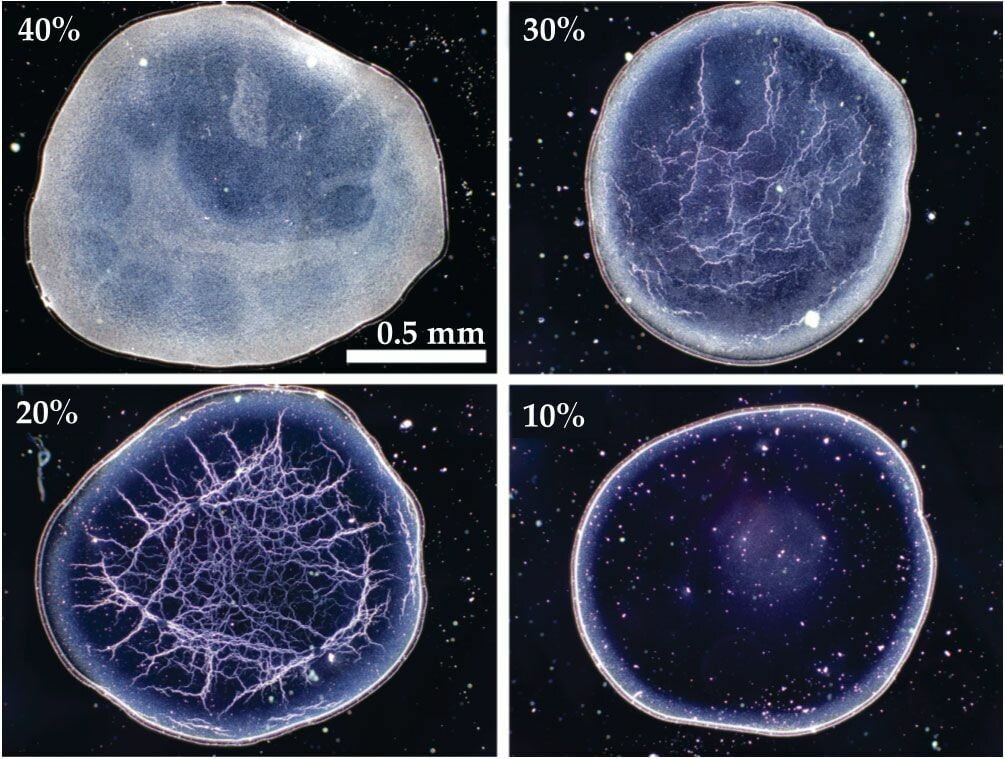
Unlike scotch whisky, when American bourbon whiskeys are diluted, they form unique web-like evaporation patterns. These differences arise in part from the way the liquors are aged: scotch is aged in re-used barrels, whereas bourbons require aging in a new, charred American white oak barrel*.
During aging, the whiskey picks up water-insoluble chemicals from the barrel. When water is added to the bourbon, it helps transport those insoluble components to the surface of a droplet, where they form a monolayer of fatty acid chains (Image 2; in green). As evaporation continues and the droplet gets smaller, the molecules at the shrinking surface collapse inward, forming the rigid web structure we see left behind. The patterns that form act as a kind of fingerprint for the bourbon. Check out some of the brand-to-brand variations over at the researchers’ Whiskey Webs site. (Image and research credit: S. Williams et al.; via Physics Today)
* In case you were wondering, this is actually a legal requirement in order to be considered bourbon. Bourbons must also be made from a grain mixture that is >50% corn.

Vanishing Spirits: Aging
The necessary ingredients for scotch whisky’s evaporation patterns are alcohol, surfactants, and polymers; some of those components are absorbed during the spirit’s aging in oak casks. Photographer Ernie Button explored how long it takes for whisky to absorb enough of these chemicals by photographing the stains left by samples aged between 1 and 5 weeks in an oak cask. He found that it takes about 5 weeks for the scotch patterns to begin emerging.
The aging process for scotch and other cask-aged spirits depends on the fluid’s flow through the porous grain of the oak. Evaporation plays a significant role in the process, so the aging process differs based on environmental conditions. For example, distillers in the dry, high-altitude climate of Colorado must use climate-controlled storage, whereas Scottish distillers use a more humid natural climate to their advantage.
Another major factor in the aging process is the charred oak cask itself. Some whiskys, like American bourbon, always use a brand new barrel, whereas scotch is often aged in a previously-used cask. With older casks, absorption of molecules from the wood takes longer, which is why scotch is typically aged for much longer than some other types of whisky. (Image, research, and submission credit: E. Button; see also)

Vanishing Spirits: Rice-Based Whisky
In yesterday’s post, photographer Ernie Button showed us that barrel-aged gin can leave behind an evaporation pattern remarkably similar to scotch whisky. But even among whiskys not every spirit uses the same grains.
Here we see patterns left behind by a 10-year-aged, rice-based whisky. The stains are entirely different than those of (barley-based) scotch. The rice leaves behind stains with distinct regions, including a radially uniform rim and an interior reminiscent of satellite photos. Presumably the interaction of rice and the cask leaves the whisky with surfactants and polymers that behave rather differently than those of scotch.
It takes time for spirits to take on character from the casks they’re aged in. Tomorrow we’ll take a look at just how much aging is necessary for scotch’s patterns to emerge. (Image, research, and submission credit: E. Button; see also)

Vanishing Spirits: Gin
Photographer Ernie Button has spent years exploring the patterns left by evaporating scotch. A team of researchers found that the uniformity of scotch whisky’s stain requires three ingredients: alcohol to drive concentration gradients, surfactants to pull particulates away from the drop’s edge, and polymers to help stick particles to the glass.
Button wondered whether other spirits might produce similar patterns, and, indeed, some do. The photos above are stains left behind by evaporated gin that’s been aged for a year in oak casks. The patterns are extremely similar in appearance to those from aged scotch whiskies, suggesting that the same fluid dynamical effects are at play here, despite the difference in liquor. But do all grain spirits make these patterns? Check back tomorrow to find out. (Image, research, and submission credit: E. Button; see also)

Pearls of Mezcal
Mezcal is a traditional Mexican liquor distilled from agave. (The more commonly known tequila is actually a special type of mezcal.) As a part of the production process, distillers pour a stream of mezcal into a bowl, creating a flotilla of small bubbles called pearls. Strange as it sounds, these pearls let the distiller judge the alcohol content of the liquor! When the ratio of alcohol and water in the mixture is just right, the bubbles will have a longer lifetime before they coalesce. If there’s too little or too much alcohol, the bubbles won’t last as long. The effect depends on both the viscosity and the surface tension of the liquor, but it’s the odd way that viscosity changes in water/alcohol mixtures that creates this Goldilocks behavior. It’s a fascinating demonstration of how traditional techniques often have true scientific underpinnings. (Video credit: M. Wilhelmus et al.)

Tears of Wine
Tears of wine are caused by the Marangoni effect, in which a gradient in surface tension causes mass flow. The water in the wine has a higher surface tension than the alcohol in the wine, causing the wine to be drawn away from regions of higher alcohol concentration. #