Although Thomas Blanchard’s latest short film, “-N- Uprising”, is less overtly fluid dynamical, fluids underlie almost every aspect of it. The blossoming of flowers is often driven by osmosis and water pressure. Spiders rely on hydraulic pressure to move their limbs, and many insects first unfurl their wings by pumping hemolymph through the network of veins that lace them. Even when hidden beneath the surface, fluid dynamics is everywhere. (Video credit: T. Blanchard; via Colossal)
Month: June 2019

Urban Centers During Hurricanes
As the climate warms, many urban centers are facing stronger and more frequent storms. Some, like New York City, are using numerical simulations to better understand the interactions of their complicated urban geometries with hurricane force winds.
Above you see a simulation showing predicted wind speeds in a Lower Eastside neighborhood. The incoming wind speed (from the left) is roughly 60 m/s (~134 mph), but the speeds around and between buildings are as much as 2 times higher than that. That means that, even if a storm is Category 3 or 4, there will be areas of a neighborhood that receive sustained winds well beyond the range of a Category 5 hurricane. Urban planners need this sort of data both for devising building requirements and for understanding what storm conditions warrant mandatory evacuations for residents. (Video and image credit: X. Jiang et al.)

Transporting Droplets
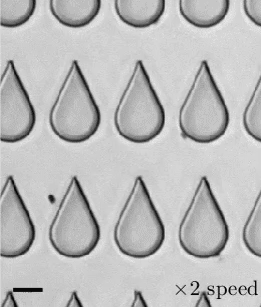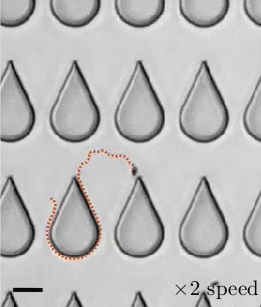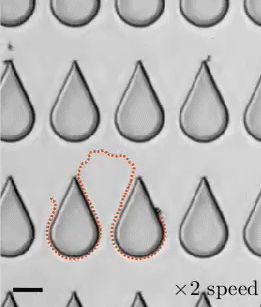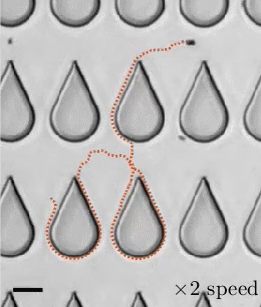
Transporting droplets easily and reliably is important in many microfluidic applications. While this can be done using electric fields, those fields can impact biological characteristics researchers are trying to measure. As an alternative, a group of researchers have developed the concept of “mechanowetting,” a technique that uses surface tension forces to hold droplets on a traveling wave.
Now visually, it’s a bit tough to see what’s going on here. In the animations, it looks like the droplets are just sticking to a moving surface, but that’s an illusion. The surface the droplet is sitting on is fixed and unmoving. It’s a thin silicone film that covers a ridged conveyor belt. The belt underneath can (and does) move. This creates a traveling wave. Instead of that wave simply passing beneath the droplet, it triggers an internal flow and restoring force that helps the drop follow the wave. The effect is strong enough that small droplets are even able to climb up vertical walls or stick upside-down. (Image, research, and submission credit: E. de Jong et al.)

Oil-on-Water Impact
Although many people have studied droplet impacts over the years, there’s been remarkably little work done with oil-on-water impacts. One of the things that makes this situation different is that the oil and water are completely immiscible, which means we can see aspects of the impact process that are invisible with, say, water-on-water impacts.
The animation above shows an underwater view of the oil droplet’s impact. The energy of the initial impact creates an expanding crater and an unstable crown splash. That crown splash contains both water and oil. After it ejects some droplets, the rim stabilizes, but we can still see small perturbations along its edge as it starts to retract. In the water, high surface tension damps out these perturbations. Not so for the oil! As the crater retracts, the small disturbances along the rim get stretched into mushroom-shaped fingers that point inward toward the impact site. Because the index of refraction is different between oil and water, we can see the fingers clearly near the end of the animation. (Image and research credit: U. Jain et al.; submitted by Utkarsh J.)

Ink Explosion
Sometimes beautiful flows come from simple combinations. Here the artists of Chemical Bouillon combine ink and hydrocarbons to create lovely explosions of color. Eschewing quick cuts between views, they allow us to linger and explore the flow ourselves as it changes. Differences in surface tension drive streaming flows along the surface, but there seem to be some chemical reactions contributing as well. Watch along the edges and you may even see convection pulling ink down and back. The whole video is only 2 minutes long and worth a full watch. (Image and video credit: Chemical Bouillon)


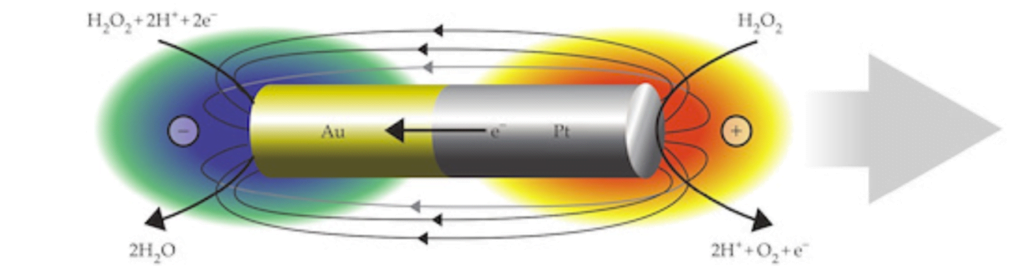
Artificial Microswimmers
In a 1959 lecture entitled “There’s Plenty of Room at the Bottom”, Richard Feynman challenged scientists to create a tiny motor capable of propelling itself. Although artificial microswimmers took several more decades to develop, there are now a dozen or so successful designs being researched. The one shown above swims with no moving parts at all.
These microswimmers are simple cylindrical rods, only a few microns long, made of platinum (Pt) on one side and gold (Au) on the other. They swim in a solution of hydrogen peroxide, which reacts with the two metals to generate a positively-charged liquid at the platinum end and a negatively-charged one at the gold end. This electric field, combined with the overall negative charge of the rod, causes the microswimmer to move in the direction of its platinum end.
Depending on the hydrogen peroxide concentration, the microswimmers can move as quickly as 100 body lengths per second, and they’re capable of hauling cargo particles with them. One planned application for artificial microswimmers is drug delivery, though this particular variety is not well-suited to that since the salty environment of a human body disrupts the mechanism behind its motion. (Image credits: swimmers – M. Ward, source; diagram – J. Moran and J. Posner; see also Physics Today)

Experimenting with Speakers
In her ongoing quest to explore natural resonance, Dianna has enlisted some very nice, very expensive speakers to find out just what happens when the bass drops. If you ever wondered what the natural frequency of your eyeballs is, then this one’s for you.
If you’re more intrigued by the idea of putting out fires with sound (and/or explosions), I’ve got some posts on that including a sound-based fire extinguisher and a supersonic cannon capable of blowing out fires. (Video credit: Physics Girl)

Floccing Particles
Adding particles to a viscous fluid can create unexpected complications, thanks to the interplay of fluid and solid interactions. Here we see a dilute mixture of dark spherical particles suspended in a layer of fluid cushioned between the walls of an inner and outer cylinder. Initially, the particles are evenly distributed, but when the inner cylinder begins to rotate, it shears the fluid layer. Hydrodynamic forces assemble the particles together into loose conglomerates known as flocs. Once the particles form these log-like shapes, they remain stable thanks to the balance between viscous drag on particles and the attractive forces that pull particles toward one another. (Image and research credit: Z. Varga et al.; submitted by Thibaut D.)

Crepe-Making Physics
If you buy a crêpe from a vendor, chances are that they’ll use a blade like the one above to spread the batter evenly across an immobile griddle. But for those of us making our own crêpes at home, this method is impractical. (After all, who wants to purchase a special griddle and utensil just for making one meal?) Instead most of us make our crêpes or pancakes in a standard pan and we use gravity to help us spread the batter.
Now researchers have described this crêpe-making process mathematically and calculated the optimal method for getting a perfect, uniformly-thin crêpe. Their model even accounts for the fact that the viscosity of the batter changes as the crêpe cooks.
For optimal crêpe-making, add the batter to the center of the pan. Then immediately tilt the pan to one side to spread the batter all the way to the edge. Keeping the pan inclined, rotate once to fill in the full circumference. Then continue the rotation at a slighter incline to fill in any holes until the pan is horizontal and the crêpe is cooked through. This is what’s shown in the lower animation, where the colormap indicates the crêpe thickness and the arrows show the effective direction of gravity. (Image credit: crêpe-making – taleitan, simulated crêpe – E. Boujo and M. Sellier; research credit: E. Boujo and M. Sellier; via APS Physics; submitted by Kam-Yung Soh)

Using Bubbles to Keep Clean
Keeping produce clean of foodborne pathogens is a serious issue, and delicate fruits and vegetables like tomatoes cannot withstand intense procedures like cavitation-based cleaning. But a new study suggests that simple air bubbles may have the power to keep our produce free of germs.
In particular, researchers studied air bubbles injected into water as they bounced and slid along an inclined solid surface. They found that as a bubble approaches a tilted surface, it squeezes a thin film of liquid between itself and the surface. That flow creates a shear stress that pushes contaminants like E. coli away from the point of impact. When the bubble bounces away, fluid gets sucked back into the void left behind, creating more shear stress. In their experiments and simulations, the team measured shear stresses greater than 300 Pa, more than double what’s needed to remove foodborne bacteria like Listeria. (Image credit: Pixabay; research credit: E. Esmaili et al.)