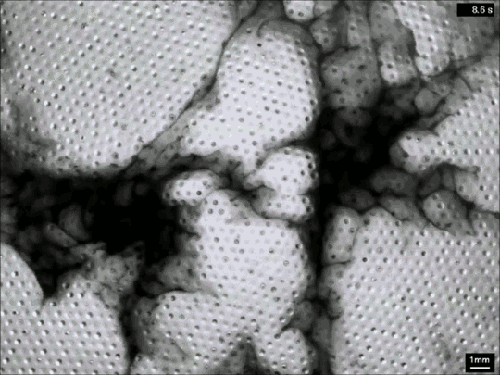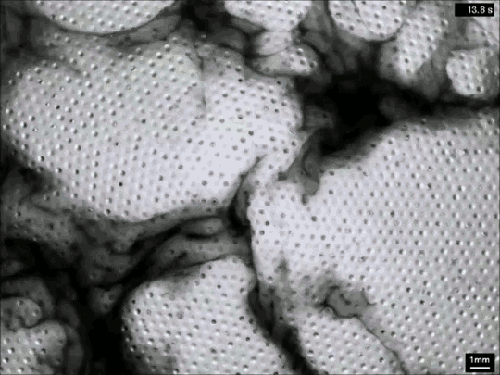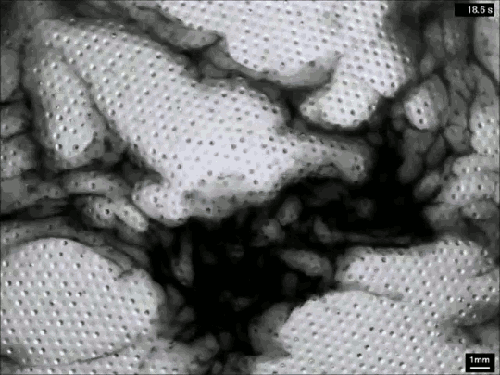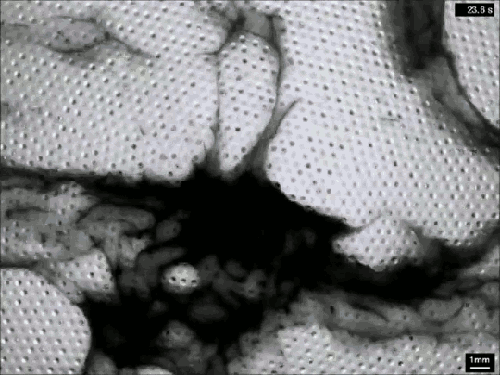
Vaucheria is a genus of yellow-green algae (think pond scum), and some species within this genus reproduce asexually by releasing zoospores. Once mature, the zoospore has to squeeze out of a narrow, hollow filament in order to escape into the surrounding fluid (top). To do so, it uses tiny hair-like flagella on its surface. Despite the minuscule size of these micron-length flagella, they generate some major flows around the zoospore (middle and bottom). Even several body lengths away, the flow field shows significant vorticity. All this active entrainment of fluid from the surroundings helps the zoospore escape its confinement and swim away to start a new plant. (Image and research credit: J. Urzay et al., source)
Tag: 2014gofm

Vortex Impact

When a vortex ring impacts a solid wall (or a mirrored vortex ring), it expands and quickly breaks up. The animations above show something a little different: what happens when a vortex ring hits a water-air interface. As seen in the side view (top image), the vortex starts to expand, but its shear at the interface generates a stream of smaller vortices that disrupt the larger vortex. (They even look like a little string of Kelvin-Helmholtz vortices!) When viewed from above (bottom image), the vortex ring impact and breakdown look even more complicated. Mushroom-like structures get spat out the sides as those secondary vortices form, and the entire structure quickly breaks up into utter turbulence. There’s some remarkable visual similarities between this situation and some we’ve seen before, like a sphere meeting a wall and drop hitting a pool. (Image credit: A. Benusiglio et al., source)

Creating Clouds
What you see here is the formation of clouds and rain – but it’s not quite what you’re used to seeing outside. This is an experiment using a mixture of sulfur hexafluoride and helium to create clouds in a laboratory. Everything is contained in a cell between two transparent plates. Liquid sulfur hexafluoride takes up about half of the cell, and when the lower plate is heated, that liquid begins evaporating and rising in the bright regions. When it reaches the cooled top plate, the liquid condenses into droplets inside the dimples on the plate, eventually growing large enough to fall back as rain. The dark wisps you see are areas where cold sulfur hexafluoride is sinking, much like in the water clouds we are used to. Setups like this one allow scientists to study the effects of turbulence on cloud physics and the formation of droplets. (Image credit: E. Bodenschatz et al., source)
Boston-area folks! I’ll be taking part in the Improbable Research show Saturday evening at 8 pm at the Sheraton Boston. Come hear about the Boston Molasses Flood and other bizarre research!

Self-Wrapping Drops
A liquid drop can fold itself up in a thin sheet. The animation above shows a drop of water with an ultra-thin (79nm) circular sheet of polystyrene atop it. As a needle removes water from the underside of the droplet, the shrinking droplet causes wrinkles and folds to form in the sheet. What’s going on here is a competition between the energy required to change the droplet’s shape and the energy needed to bend the sheet. Eventually, the droplet’s volume is small enough that the bending of the sheet overrules surface tension in dictating the droplet’s shape. The result is a tiny empanada-shaped droplet completely encapsulated by the sheet. (Image credit: J. Paulsen et al., source; research paper)

Pearls of Mezcal
Mezcal is a traditional Mexican liquor distilled from agave. (The more commonly known tequila is actually a special type of mezcal.) As a part of the production process, distillers pour a stream of mezcal into a bowl, creating a flotilla of small bubbles called pearls. Strange as it sounds, these pearls let the distiller judge the alcohol content of the liquor! When the ratio of alcohol and water in the mixture is just right, the bubbles will have a longer lifetime before they coalesce. If there’s too little or too much alcohol, the bubbles won’t last as long. The effect depends on both the viscosity and the surface tension of the liquor, but it’s the odd way that viscosity changes in water/alcohol mixtures that creates this Goldilocks behavior. It’s a fascinating demonstration of how traditional techniques often have true scientific underpinnings. (Video credit: M. Wilhelmus et al.)