“Liquid Skies” by Roman De Giuli is full of colorful but nebulous fluid imagery. The visuals consist of liquids like paint, ink, and alcohol filmed in macro atop paper. You can catch a behind-the-scenes glimpse of De Giuli at work here. (Image and video credit: R. De Giuli)
Year: 2021

Acidic Sea Spray
As waves crash and break, they generate a spray of droplets — known as aerosols — that make their way into the atmosphere. Researchers investigated the chemistry of these aerosol droplets by generating spray in a wave tank filled with ocean water. They found that aerosol droplets are far more acidic than the ocean they come from, and the smaller the droplet, the more acidic it is. This acidification happens in a matter of minutes, as acidic gases interact with the spray. Their findings will be critical for accurately modeling the climate connections between our oceans and atmosphere. (Image credit: Elle; research credit: K. Angle et al.; via OceanBites; submitted by Kam-Yung Soh)

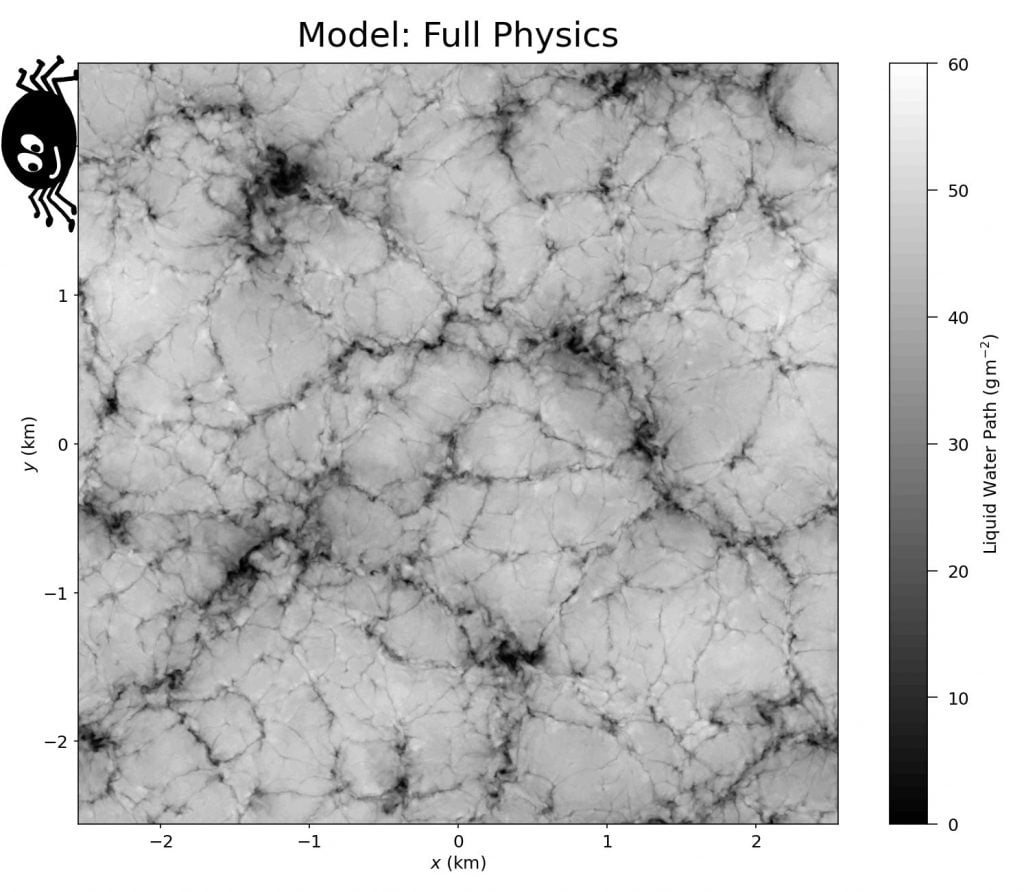
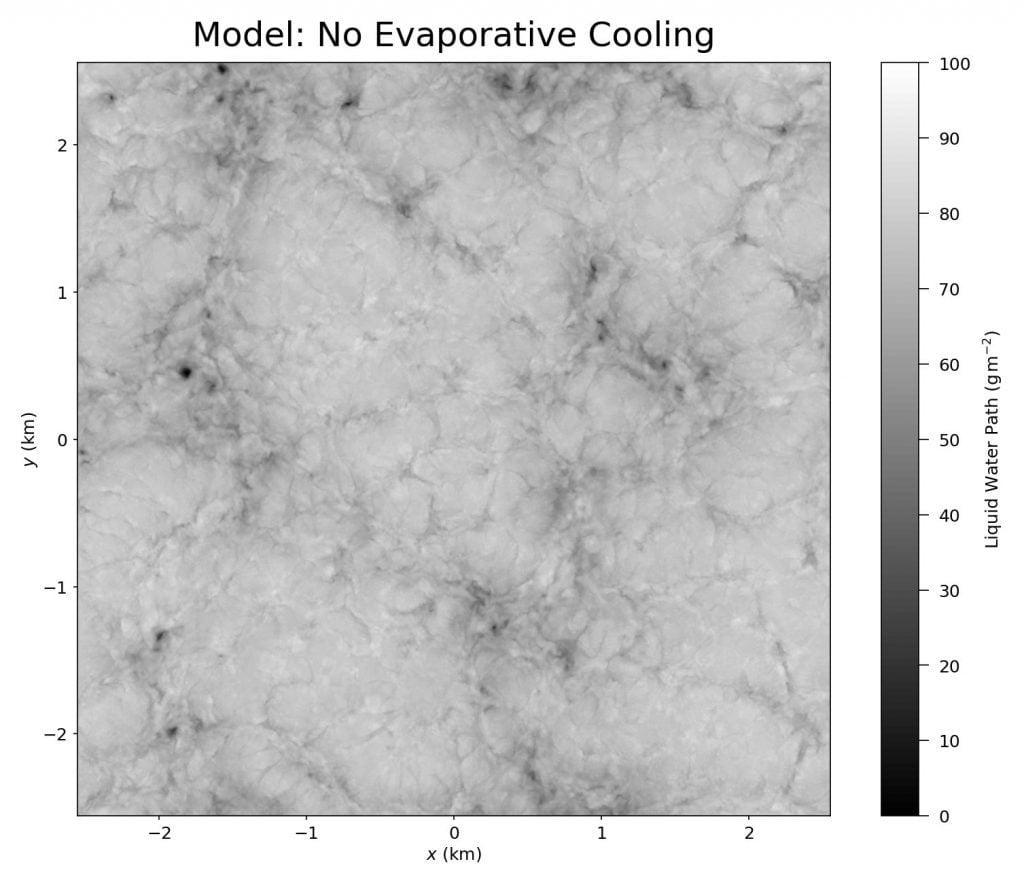
Spiderwebs and Stratocumulus Clouds
Stratocumulus clouds cover about 20% of Earth’s surface at any given time, and they form distinctive patterns of lumpy cells separated by thin slits. Because of their interconnectedness, researchers nicknamed these narrow regions spiderwebs. New simulations show that evaporative cooling along the cloud tops drives the formation of these spiderwebs (Image 2). Without it (Image 3), the cloud pattern looks very different. (Image credits: featured image – L. Dauphin/MODIS, others – UConn ME 3250; research credit: G. Matheou et al.)

The Unsinkable Pygmy Gecko
The Brazilian pygmy gecko is a tiny inhabitant of the Amazon rainforest, growing to no longer than 24 mm. But these tiny lizards have some incredible superpowers when it comes to surviving the rainforest’s deluges. The gecko’s surface is superhydrophobic — water repellent — thanks to millions of tiny hairs that create air pockets between water and the gecko’s skin. This superhydrophobic surface, combined with the gecko’s tiny stature, allow it to sit atop water, supported entirely by surface tension. (Image and video credit: BBC Earth)

The Fluidity of Worm Blobs
The aquatic blackworm forms blobs composed of thousands of individual worms for protection against evaporation, light, and heat. The worms braid themselves together (Image 1). Once a blob forms, it is extremely viscoelastic, displaying properties both solid and fluid in nature (Image 2).
The worm blobs act like a collective; they bunch up to prevent evaporation that would desiccate the worms. Under intense light, the blob contracts (Image 3). The worms also prefer colder temperatures (again, to prevent evaporation) and will move toward the colder side of a temperature gradient. Under dim light, they’ll move individually, but in brighter light, the worms move collectively as a blob (Image 4).
To do so, worms on the colder side of the blob pull toward the cold, whereas worms elsewhere in the blob wiggle (Image 5). Their wiggling helps lift the blob and reduce its friction so that the pulling worms can move the blob in the right direction. For more, check out this excellent thread by one of the authors. (Image and research credit: Y. Ozkan-Aydin et al.; via S. Bhamla; submitted by Maximilian S.)

“Flux Capacitor”
Sandro Bocci’s short film “Flux Capacitor” explores the geometry and dynamics of soap films. When you dip wire models into soapy solution, the films that cling to the model can form complicated shapes as surface tension works to minimize the overall surface area. Bocci’s macro photography highlights the intense flows going on in the narrow regions where films meet. It’s a different take on soap films and neat to see! (Image, video, and submission credit: S. Bocci et al.)

Gathering Droplets
In deserts around the world, plants have adapted to collect as much moisture as they can. Geometry aids them in this endeavor because droplets on the tip of a cone will move toward its thicker base. The motion takes place due to a imbalance in surface tension forces on either end of the droplet.
As the droplet moves up a cone, it changes shape from a barrel-like drop that fully covers the conical surface to a clamshell-shaped droplet that hangs only from the bottom of the cone. (Image and research credit: J. Van Hulle et al.)

Jellyfish Make Their Own Walls
When we walk, the ground’s resistance helps propel us. Similarly, flying or swimming near a surface is easier due to ground effect. Most of the time swimmers don’t get that extra help, but a new study shows that jellyfish create their own walls to get that boost.
Of course, these walls aren’t literal, but fluid dynamically speaking, they are equivalent. Over the course of its stroke, the jellyfish creates two vortices, each with opposite rotation. One of these, the stopping vortex, lingers beneath the jellyfish until the next stroke’s starting vortex collides with it. When two vortices of equal strength and opposite rotation meet, the flow between them stagnates — it comes to halt — just as if a wall were there.
In fact, mathematically, this is how scientists represent a wall: as the stagnation line between a real vortex and a virtual one of equal strength and opposite rotation. It just turns out that jellyfish use the same trick to make virtual walls they can push off! (Image and research credit: B. Gemmell et al.; via NYTimes; submitted by Kam-Yung Soh)

Coastal Erosion
The same dynamic forces that make coastlines fascinating create perennial headaches for engineers trying to maintain coastlines against erosion. This Practical Engineering video discusses some of the challenges of coastal erosion and how engineers counter them.
In a completely undeveloped coastline, waves and storms erode the shoreline while rivers and currents replenish sand through sedimentation. Manmade structures tend to strengthen erosion processes while disrupting the sedimentation that would normally counter it. Beach nourishment — where sand gets dredged up and deposited on a beach — is an engineered attempt to replace natural sedimentation.
Dunes, mangrove forests, and wetlands are all nature’s way of protecting and maintaining coastlines. We engineers are still learning how to both utilize and protect shorelines. (Image and video credit: Practical Engineering)

Why Food Sticks to Nonstick Pans
Whether you’re cooking with ceramic, Teflon, or a well-seasoned cast iron pan, it seems like food always wants to stick. It’s not your imagination: it’s fluid dynamics.
As the thin layer of oil in your pan heats up, it doesn’t heat evenly. The oil will be hotter near the center of the burner, which lowers the surface tension of the oil there. The relatively higher surface tension toward the outside of the pan then pulls the oil away from the hotter center, creating a hot dry spot where food can stick.
To avoid this fate, the authors recommend a thicker layer of oil, keeping the burner heat moderate, using a thicker bottomed pan (to better distribute heat), and stirring regularly. (Image and research credit: A. Fedorchenko and J. Hruby)