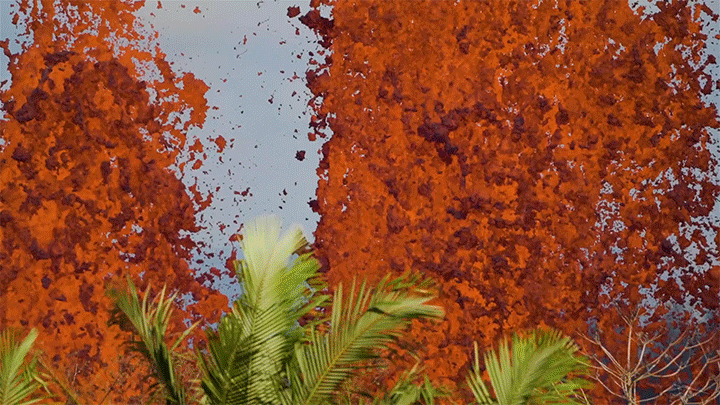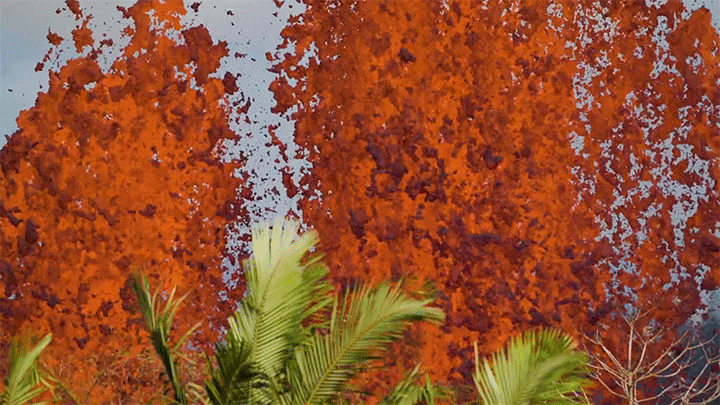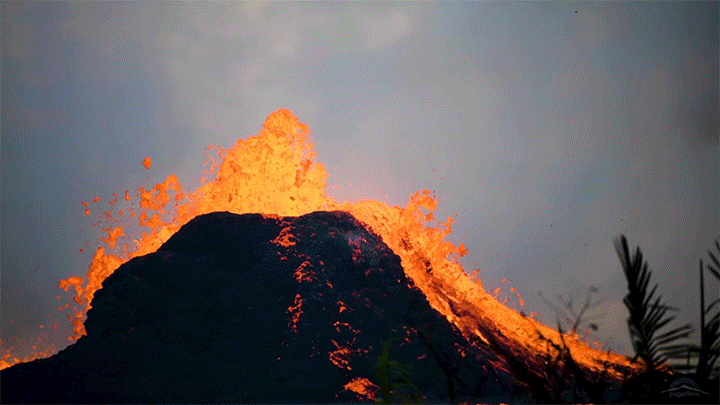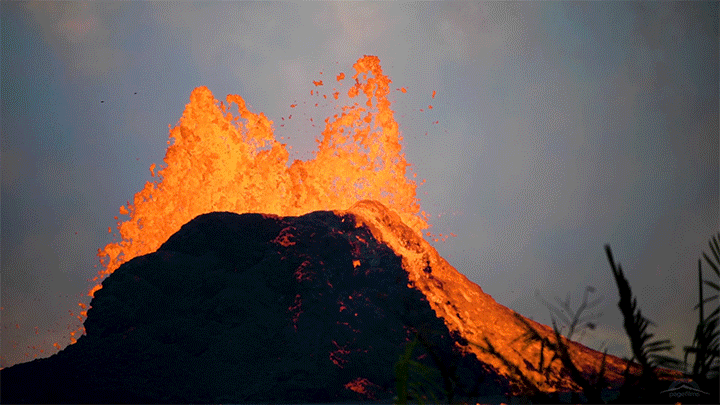The 2018 eruption of Kilauea was a dramatic example of nature’s power. This short film shows both some familiar views of that eruption as well as new ones. I found the slow-moving wall of cooling a’a lava eating the forest particularly intriguing, not least thanks to the glass-like sound of the lava advancing. Whether slow-moving or fast, lava’s destructive power is incredible to watch. (Video and image credit: Page Films)
Month: April 2020

Crisscrossing Wave Clouds
Crisscrossing lines of wave clouds mark the wake of the Sandwich Islands in this satellite image. The tallest islands in the chain thrust rocky peaks more than 1000 meters above sea level, disrupting winds flowing across the ocean. Incoming air is forced up and over the mountain, which puts it at odds with the surrounding air at that height.
Due to differences in temperature and density, the disrupted air will continue to rise and sink periodically as it flows onward. At some heights it will cool enough to condense its water vapor into clouds, and at others, it will warm enough to lose any cloud cover. This is what creates the bands of clouds we see behind each individual island. (Image credit: L. Dauphin/NASA; via NASA Earth Observatory)

COVID-19 and Outdoor Exercise
By now you’ve probably come across some blog posts and news articles about a new pre-print study looking at the aerodynamics of running and the potential exposure to exhaled droplets. And you may also have seen articles questioning the accuracy and validity of such simulations. I’ve had several readers submit questions about this, so I dug into both the research and the criticisms, and here are my thoughts:
Is this study scientifically valid?
I’ve seen a number of complaints that since this paper hasn’t been peer-reviewed, we shouldn’t trust anything about it. That seems like an unreasonable overreaction to me considering how many studies receive press attention prior to their actual peer-reviewed publication. This is not a random CFD simulation produced by someone who just downloaded a copy of ANSYS Fluent. This work comes from a well-established group of engineers specializing in sports aerodynamics, and long-time readers will no doubt recognize some of their previous publications. Over the past decade, Blocken and his colleagues have become well-known for detailed experimental and simulation work that indicates larger aerodynamic effects in slipstreams than what we generally recognize.
In this paper, they lay out previous (biological) studies related to SARS and droplet exhalation; they use those papers and several wind tunnel studies to validate computational models of droplet evaporation and runner aerodynamics; and then they use those inputs to simulate how a cloud of exhaled droplets from one runner affects someone running alongside, behind, or in a staggered position relative to the first runner.
In other words, their work includes all the components one would expect of a scientific study, and it makes scientifically justifiable assumptions with regard to its methods. (That’s not, mind you, to say that no one can disagree with some of those choices, but that’s true of plenty of peer-reviewed work as well.) All in all, yes, this is a scientifically valid study, even if it has not yet undergone formal peer-review*.
Can simulations actually tell us anything about virus transmission?
One complaint I’ve seen from both biologists and engineers is that simulations like these don’t actually capture the full physics and biology involved in virus transmission. While I agree with that general sentiment, I would point out two important facts:
1) Blocken et al. acknowledge that this is not a virology study and confine their scientific results to looking at what happens physically to droplets when two people are moving relative to one another. Whether those droplets can transmit disease or not is a question left to biological researchers.
2) Most medical and biological research also does not account for the physics of droplet transmission and transport. For the past century, this research has focused almost exclusively on droplet sizes, with the assumption that large droplets fall quickly and small droplets persist a little longer. To my knowledge, some of the only work done on the actual physics of the turbulent cloud produced by coughing or sneezing comes from Lydia Bourouiba’s lab at MIT. And, to me, one of the fundamental conclusions from her work is that droplets (especially small ones) can persist a lot longer and farther than previously assumed. Can those droplets facilitate transmission of COVID-19? The general consensus I’ve seen expressed by medical experts is no, but, to my knowledge, that is based on opinion and assumption, not on an actual scientific study.
The bottom line
In my opinion, there’s a big disconnect right now between the medical/biological community and the engineering community. To truly capture the physics and biology of COVID-19 transmission requires the expertise and cooperation of both. Right now both sides are making potentially dangerous assertions.
Honestly, based on what I know about aerodynamics, I am personally skeptical as to whether 6 ft of physical separation is truly enough; whether it is or not seems to depend on how transmissible the novel coronavirus is through small droplets, which, again, to my knowledge, is unestablished.
Should we leave more distance than 6ft between us when exercising outdoors? Absolutely. Aerodynamically, it makes perfect sense that following in someone’s slipstream would put you inside their droplet cloud, which needs time and space to disperse. Personally, I’ve sidestepped the question entirely by doing all my cycling indoors while quarantined.
tl;dr: There are a lot of open questions right now about COVID-19 transmission and what qualifies as safe distancing, but it’s smarter to err on the side of more distancing. Don’t hang close to others when running or cycling outdoors.
(Image and research credit: B. Blocken et al.; submitted by Corky W. and Wendy H.)
*I will add that, with my training, I have and do occasionally peer-review studies such as this one, and I read the full paper with the same sort of critical eye I would turn to a paper I was asked to review.

Watching a Droplet Freeze
Whether it’s rain hitting an airplane wing or droplet-based 3D printing, the dynamics of a droplet impacting and solidifying on a surface are important. This new study observes the process from below, tracking the progress of freezing on a scale of hundreds of nanoseconds.
All three of the drops you see above are liquid hexadecane. Each droplet was the same size and impacted at the same velocity. What differs in each image is how much colder the surface was than hexadecane’s melting point. The leftmost image shows a droplet on a surface only a few degrees cooler than the melting point. The initial expanding ring shows the droplet’s contact line expanding as it impacts. Then frozen crystals appear and grow inside the drop until the entire thing freezes.
With a slightly colder surface (middle image), frozen crystals form while the contact line is still expanding, and rather than form in distinctive spots, they form as a cloud that quickly expands throughout the drop.
But with an even colder surface (right image), something entirely new happens. As the drop freezes, we see multiple dark rings expand through the drop. Each of these rings is made up of frozen crystals. The researchers argue that we’re seeing a combination of freezing and hydrodynamics here. Essentially, whenever the frozen crystals get large enough, the outward flow of the impacting drop sweeps them toward the contact line. As new crystals grow near the center of the drop, they’re dragged out in a subsequent wave. (Image, research, and submission credit: P. Kant et al.)

Tektites and Spinning Fluids
Tektites, like obsidian, are a naturally-occurring glass formed from molten rock. But tektites are often dumbbell or figure-8-shaped because they form in midair from spinning bits of fluid sent skyward after the crash of a meteor. In this video, Steve Mould takes us through the process and discusses some recent work by scientists who’ve created artificial tektites in the lab by levitating and spinning candle wax and other fluids. (Video and image credit: S. Mould; research credit: K. Baldwin et al.)

“Focus, Vol. 1”
In “Focus, Vol. 1,” photographer Roman De Giuli follows colorful droplets as they roll along, chase one another, and burst. You may notice that many of the drops seem attracted to one another. This is actually a surface tension effect caused by the dimples the droplets create on the surface; it’s the same effect responsible for Cheerios clumping together in your milk. Interestingly, though, the oil coating the drops doesn’t seem to drain quickly enough for the clumping drops to actually coalesce. (Image and video credit: R. De Giuli)

Why Animals Shake Themselves Dry
For many animals, letting themselves air-dry is not an option. They would become hypothermic before their wet fur dried completely. This is why dogs and many other furry mammals shake themselves dry. It’s a remarkably efficient process, too, removing the majority of water from fur in a matter of seconds.
The key is to shake at a frequency such that the centrifugal force of the shake overcomes surface tension’s ability to keep the water attached to fur. The looseness of a dog’s skin (compared to humans!) is a bonus for them; the extra translation as they shake increases the centrifugal force, allowing them to shed more water more quickly. (Image and video credit: BBC Earth; research credit: A. Dickerson et al.)

Bouncing Off Hydrophilic Surfaces
Droplets typically bounce off hydrophobic surfaces due to air trapped beneath the liquid that prevents contact between the drop and surface. But even extremely smooth, hydrophilic surfaces can elicit a bounce under the right circumstances, as shown in a new study.
The key is that the droplet must bounce at exactly the right speed. If the bounce has too much momentum, it will squeeze the nanometer-sized air cushion too thin, allowing contact. Too slow and the Van der Waals attraction between the droplet molecules and wall molecules will have time to act. But between those lies a sweet spot where the dimple and cushion of air beneath the drop keep it from impacting. (Image credit: droplets – klickblick, drop bounce – J. Kolinski, bounce sim – J. Sprittles et al.; research credit: M. Chubynsky et al.; submitted by James S.)

Why Compressed Air Cans Get Cold
Anyone who’s used a can of compressed air to clean their computer or keyboard knows that the can quickly gets quite cold to the touch. This Minute Physics video explores some of the thermodynamics behind that process. Henry first identifies a few explanations that don’t quite line up with observations, before focusing in on the contents of the can: 1,1-difluoroethane. Inside the sealed can, this chemical sits in an equilibrium of part-liquid, part-vapor. But when pressure is released by opening the nozzle, the liquid boils, generating extra vapor and cooling whatever remains in the reservoir.
Although it’s not a good explanation for the compressed air can’s cooling, the cooling of an expanding gas is very important in applications like supersonic wind tunnels. That first equation you see at 0:36 in the video (for isentropic adiabatic expansion) is key to what happens in a nozzle with supersonic flow. As the flow accelerates to supersonic speeds, its temperature drops dramatically. When I was in graduate school, we actually had to preheat our hypersonic wind tunnel (in pretty much the same way you would preheat your oven at home) before we ran at Mach 6 because otherwise the temperature inside the test section would drop so low that the oxygen would liquefy out of the air! (Image and video credit: Minute Physics)

The Birth of a Liquor
A water droplet immersed in a mixture of anise oil and ethanol displays some pretty complicated dynamics. Its behavior is driven, in part, by the variable miscibility of the three liquids. Water and ethanol are fully miscible, anise oil and ethanol are only partially miscible, and anise oil and water are completely immiscible. These varying levels of miscibility set up a lot of variations in surface tension along and around the droplet, which drives its stretching and eventual jump.
Once detached, the droplet takes on a flattened, lens-like shape that continues to spread. That spreading is driven by the mixing of ethanol and water, which generates heat and, thus, convection around the drop. This not only spreads the droplet, it causes turbulent behavior along the drop’s interface. (Image and video credit: S. Yamanidouzisorkhabi et al.)