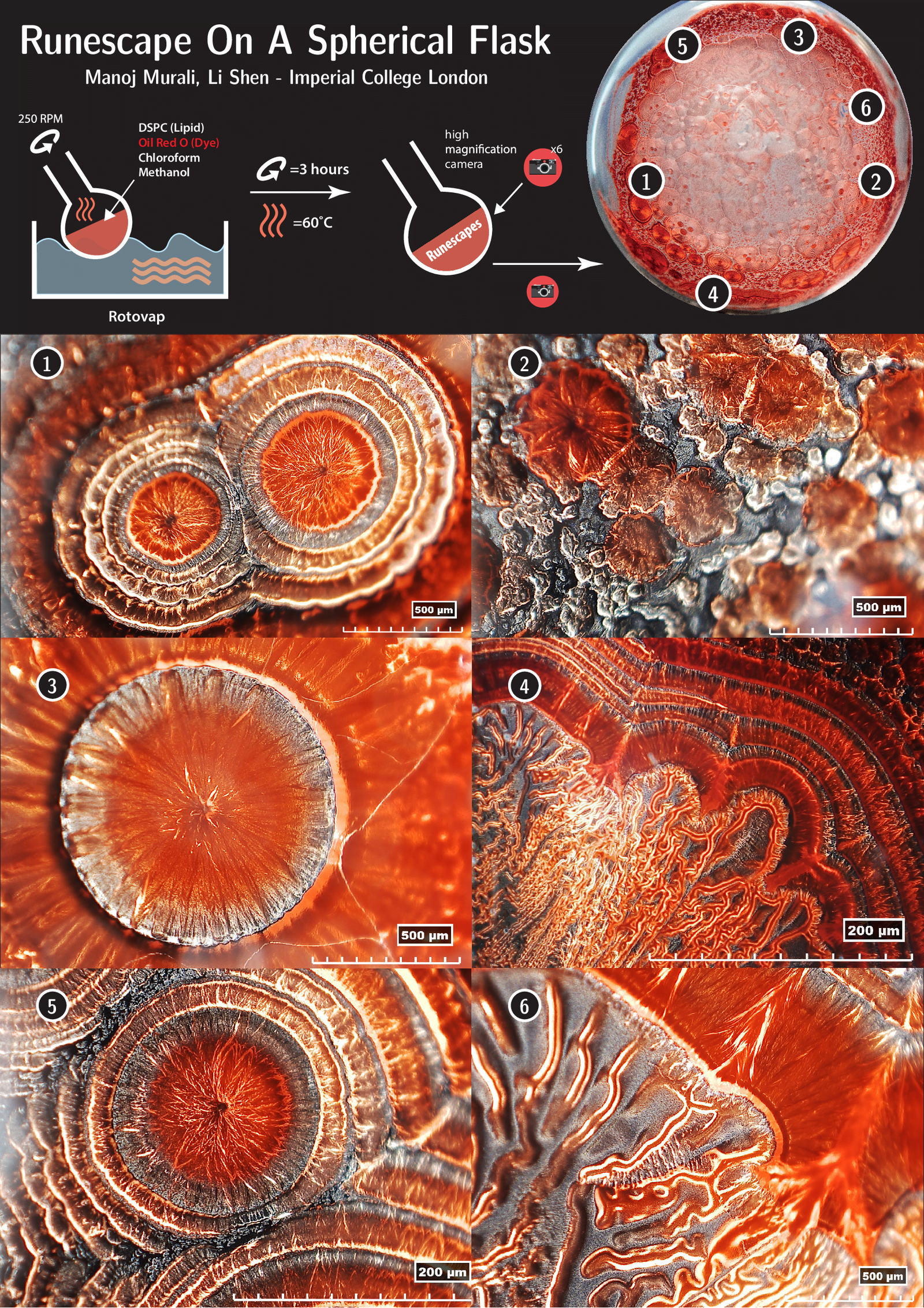
Drying fluids can leave behind all kinds of fascinating patterns, as we’ve seen before with whiskey, coffee, and even blood. Here researchers study patterns left behind by lipids, dyes, and other fluids. They place their mixture in a rotating flask kept in a warm bath. For a few hours, the fluids mix, chemically react, and evaporate. The complex interactions that take place in that time leave behind fascinating, rune-like patterns, seen here under a microscope. It’s a bit like looking at photos of Martian landscapes! (Image credit: M. Murali and L. Shen)
Tag: reaction-diffusion system

Envisioning Chemical Patterns
One of the most beautiful chemical reactions is the Belousov–Zhabotinsky reaction, and it’s the subject of the latest video from Beauty of Science. Known colloquially as the BZ reaction, it begins as growing spots of color that turn into rings and chaotic spirals as the chemical reaction progresses.
Mathematically, the BZ reaction is a type of reaction-diffusion system, meaning that the patterns we see depend both on the speed of local chemical reactions and the time necessary for chemical reagents to move (or diffuse) throughout the dish. Although the diffusion in these systems can simply be the random wandering of molecules, fluid dynamics also plays a role. Variations in chemical concentration between different regions of the reaction drive fluid flows that continue to feed the pattern-making until all the reagents are consumed. (Image and video credit: Beauty of Science; via PetaPixel; submitted by clogwog)