As a bubble sitting on a pool collapses, wrinkles form around its edges. Visually, the result is quite similar to the wrinkles one gets on an elastic sheet. Unlike the solid sheet, though, the bubble’s film varies in thickness; we know this because of the fringes shown in the enlarged inset of the poster. Researchers are studying this non-uniformity to see whether it affects the number and shape of wrinkles that form on the bubble. (Image and research credit: O. McRae et al.)
Tag: physics

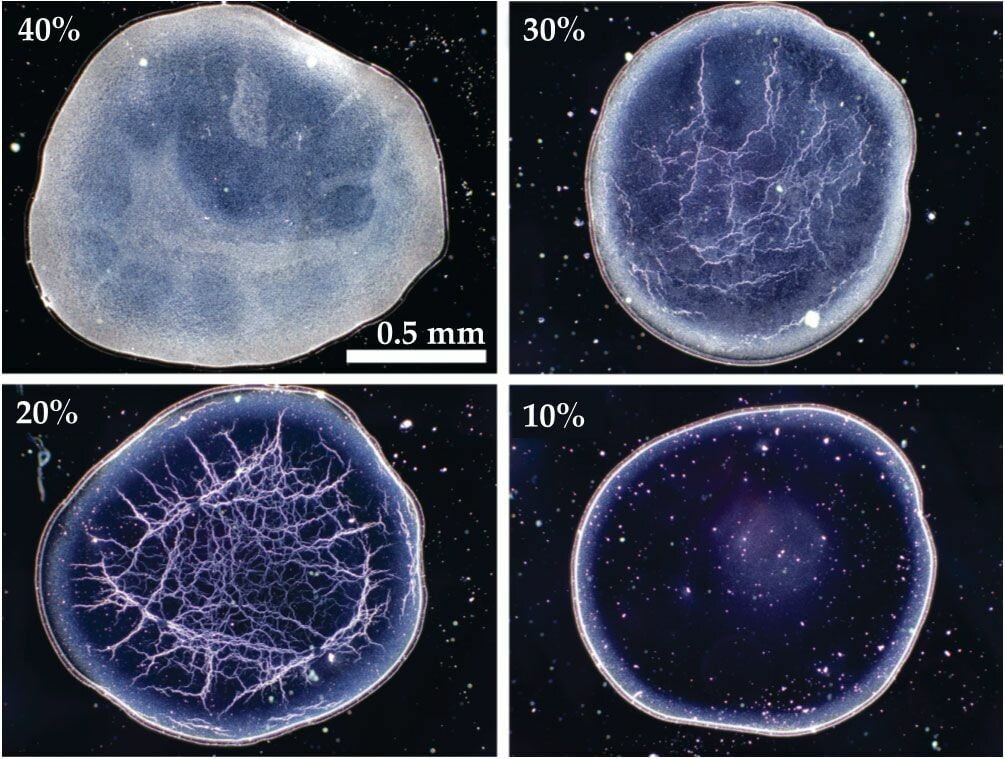
Whiskey Webs
Unlike scotch whisky, when American bourbon whiskeys are diluted, they form unique web-like evaporation patterns. These differences arise in part from the way the liquors are aged: scotch is aged in re-used barrels, whereas bourbons require aging in a new, charred American white oak barrel*.
During aging, the whiskey picks up water-insoluble chemicals from the barrel. When water is added to the bourbon, it helps transport those insoluble components to the surface of a droplet, where they form a monolayer of fatty acid chains (Image 2; in green). As evaporation continues and the droplet gets smaller, the molecules at the shrinking surface collapse inward, forming the rigid web structure we see left behind. The patterns that form act as a kind of fingerprint for the bourbon. Check out some of the brand-to-brand variations over at the researchers’ Whiskey Webs site. (Image and research credit: S. Williams et al.; via Physics Today)
* In case you were wondering, this is actually a legal requirement in order to be considered bourbon. Bourbons must also be made from a grain mixture that is >50% corn.

Albedo Effect
Temperature isn’t the only factor that determines how ice will melt. In this photo, a dark oak leaf absorbed more solar radiation than the reflective ice around it, causing the ice beneath it to melt. Scientifically, this effect is described by albedo; darker, more absorptive surfaces like the leaf have a lower albedo, whereas light, reflective ice and snow have a high albedo and can better resist melting on sunny days. (Image credit: K. James; submitted by Kam-Yung Soh)

Visualizing Radiation
Radiation is invisible, but it’s not too difficult to build an apparatus that lets you see it. This video shows the ghostly aftermath of passing radiation in a cloud chamber, one of the first set-ups used to study radiation. The chamber contains a radioactive source and chilled isopropyl alcohol. The alcohol forms a supersaturated vapor — essentially a cloud in waiting — inside the chamber.
When a radioactive particle gets emitted from the source, it streaks through the chamber, colliding with atoms and ionizing them. Those ions then serve as nucleation sites where alcohol condenses into droplets. It’s these condensation trails that we see bloom and decay in the particle’s wake. (Image and video credit: L. Gledhill)

Insect-Inspired Flight
Insects are incredibly agile and resilient fliers, capable of colliding and recovering without damage. Engineers are only beginning to capture these characteristics in their robots. Here, engineers use a soft actuator — a rubber cylinder coated in carbon nanotubes — to drive their robot’s flight. When voltage is applied across the carbon nanotubes, the rubber squeezes and stretches, causing the robot’s wings to flap. These soft actuators are far less fragile than hard ones, allowing the robots to take hits and keep flapping, much like the real insects. (Image and video credit: MIT News; research credit: K. Chen et al.)

Fluid Chains
In this video, Steve Mould tackles a question many of us have likely wondered: just why does falling water make this chain-like shape? When pouring from a slit-like orifice, water jets take on this undulating pattern. While I have no issue with Steve’s explanation of surface tension oscillations driving the shape, I’ll quibble a little bit with the idea that this hasn’t been studied. Personally, I’d connect it to the fishbone instability, which classically occurs when two jets collide. At low flow rates, though, the colliding jets form a pattern very much like this one. And if you look just past the initial conditions at the container opening, all of these flows have thicker jet-like rims colliding. I think the flows in these videos are just a slightly messier version of the low-flow-rate fishbone. What do you think? (Video and image credit: S. Mould)

Meltwater Tracking Via Seal
Monitoring meltwater from Antarctic glaciers is critical for understanding our changing climate, but such remote and inaccessible regions are tough to collect data in. So researchers are turning to local workers to help them gather data. By collecting and analyzing data from seal tags, researchers have mapped new seasonal variations in meltwater flows around Pine Island Glacier. Although the seals are somewhat tough collaborators — they rarely swim exactly where the researchers would like them to — their winter activities are revealing data researchers could never have collected on their own. (Image credit: Y. Rzhemovskiy; research credit: Y. Zheng et al.; via Gizmodo)

Superior Mirage
This photograph of a ship seemingly floating far above the water is not some Photoshop fakery; it’s physics creating the illusion. It’s an example of what’s known as a superior mirage — superior because the mirage appears above the object’s actual location, unlike the mirages you see above the road on a hot day.
In this case, the air layer near the water is cold — colder than the air above it, thanks to a temperature inversion. Cold air is denser and has a higher index of refraction, so light traveling through it gets bent downward. To a far off observer, this downward bend makes objects appear higher in altitude than they actually are. The effect is most common in polar regions, where the right conditions can actually allow images of objects completely below the horizon! (Image credit: D. Morris; via The Guardian; submitted by Alec)

“Dancing With Danger”
Filmmaker Chris Bryan captures surfer Kipp Caddy as he rides an enormous wave in “Dancing With Danger.” Nothing quite captures the majesty of these powerful flows like high-speed videography. Enjoy the break, the spray, and those awesome rib vortices. (Image and video credit: C. Bryan)

Jumping Frost
Liquid water is easily electrically charged, due to its polar nature. That’s why rubbing a comb is enough to deflect a stream of water. Ice is harder to charge, but it can happen, especially when there are temperature gradients across the ice.
That’s the key behind this study of jumping frost. When ice crystals grow on a surface much colder than their surroundings, positive charges gather in the colder region, leaving the dendritic branches of the ice negatively charged. When researchers brought liquid water near the charged ice crystals, the water became charged, too. Positive charges in the water attracted the negatively-charged dendrites, causing the ice crystals to jump off the surface.
Studies like this help us better understand cloud and rain formation and may one day lead to new ways of de-icing surfaces. (Image credit: frost – Miriams-Fotos, figure – R. Mukherjee et al.; research credit: R. Mukherjee et al.; via ChemBites; submitted by Kam-Yung Soh)