Popcorn’s explosive pop looks pretty cool in high-speed video, but just watching it with a regular camera doesn’t show everything that’s going on. If we take a look at it through schlieren optics, the kernel’s pop looks even more extraordinary:
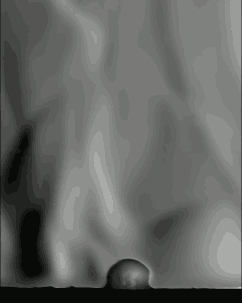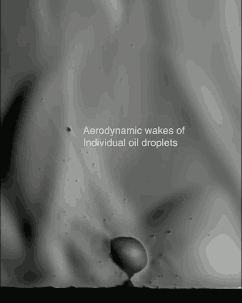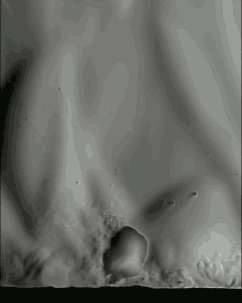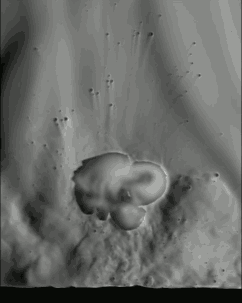
The schlieren technique reveals density differences in the gases around the corn–effectively allowing us to see what is invisible to the naked eye. The popcorn kernel acts like a pressure vessel until the expansion of steam inside causes its shell to rupture. The first hints of escaping steam send droplets of oil shooting upward. The kernel may hop as steam pours out the rupture point, causing the turbulent billowing seen in the animation above. As the heat causes legs of starch to expand out of the kernel, they can push off the ground and propel the popcorn higher. As for the eponymous popping sound, that is the result of escaping water vapor, not the actual rupture or rebound of the kernel! See more of the invisible world surrounding a popping kernel in the video below. (Image credits: Warped Perception, source; Bell Labs Ireland, source; WP video via Gizmodo; BLI video submitted by Kevin)
