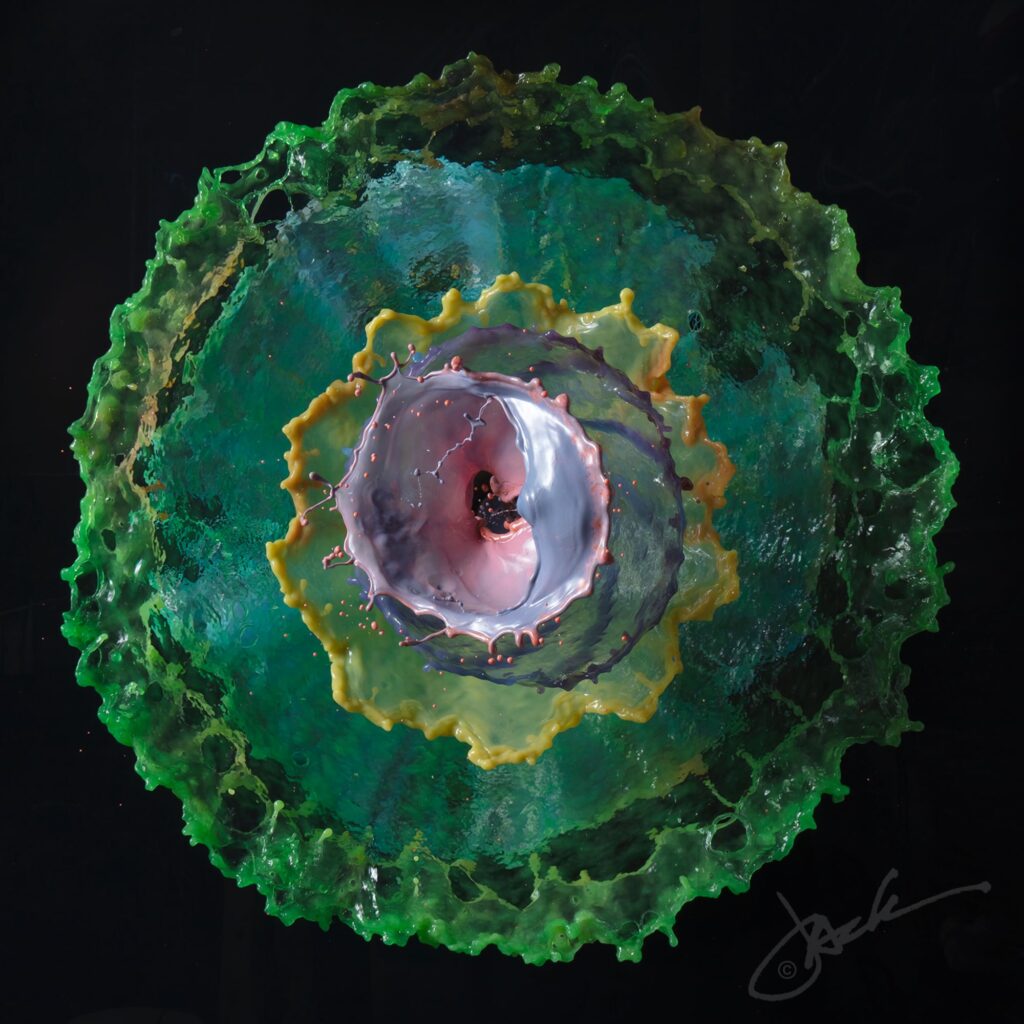
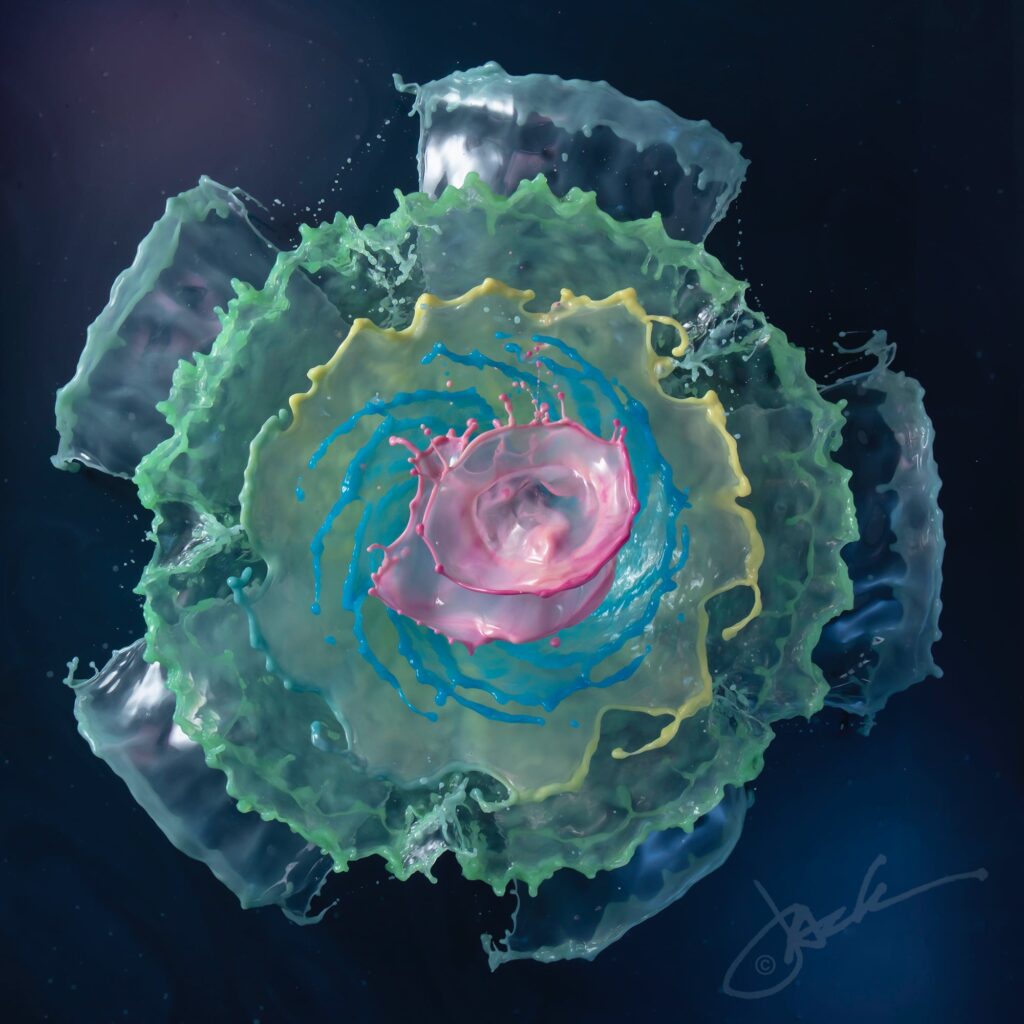
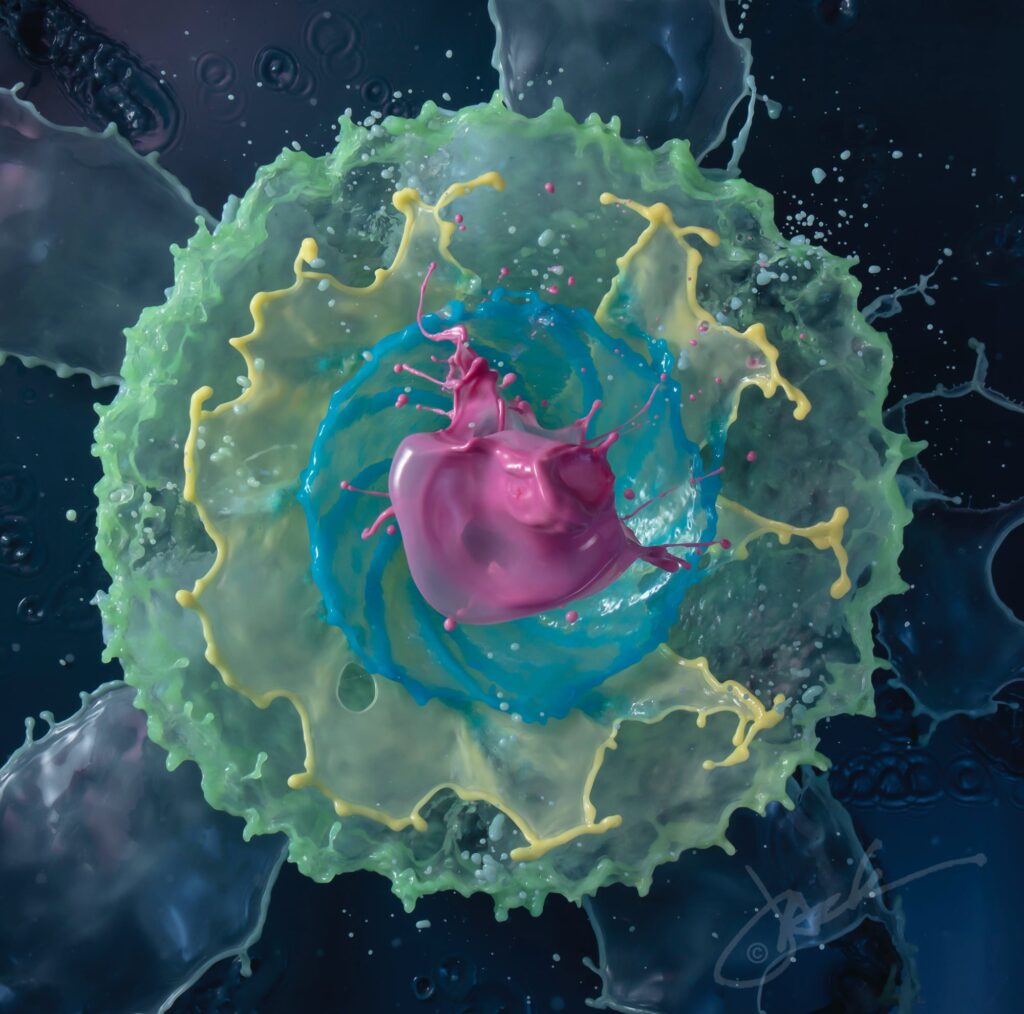
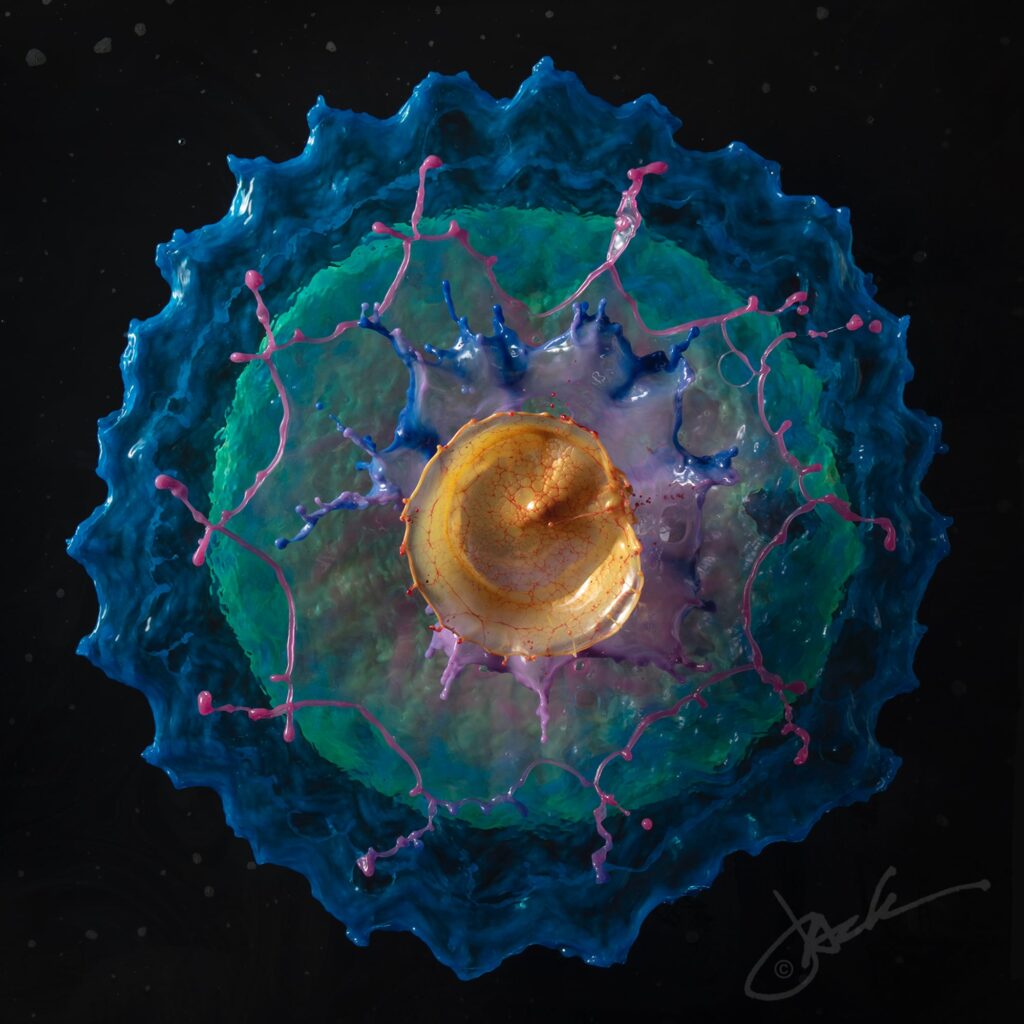
Concentric circles of colorful water float in the frame of photographer Jack Long’s images. At first glance, the liquid sculptures appear to be the splashes from one or more falling objects. But, in fact, Long reports to Colossal that the water burbles up from a custom-designed fountain. The effect is a very neat one, and I love examining the details of Long’s images. The rim of each ring is visibly thickened and often wavy in a regular pattern, hinting at an underlying Plateau-Rayleigh instability driving the inevitable break-up. Find more of Long’s work at his website and on Instagram. (Image credit: J. Long; via Colossal)
Month: December 2022

To Fizz or Not to Fizz
Place a drop of carbonated water on a superhydrophobic surface and it will slide almost frictionlessly, much the way Leidenfrost drops do. The drop behaves this way thanks to the self-produced layer of carbon dioxide vapor that it levitates on. As the gas escapes, the drop eventually settles back into contact with its surface. But until then, its levitation makes for some fun.

On the treated half of the glass (left), bubbles form a continuous film against the glass. On the untreated side (right), bubbles nucleate, grow, and rise as expected for a fizzy drink. Single droplets aren’t the only source of fun, however. In the images above, researchers coated the left half of a wine glass with a superhydrophobic treatment, while leaving the right half of the glass untouched. Once (dyed) carbonated water is poured into the glass, we see a bizarre dichotomy. In the right, untreated half of the glass, carbon dioxide bubbles nucleate, grow, and rise through the glass. But on the left side, the liquid appears still and bubble-less. In fact, the carbon dioxide gas on the left side is forming a continuous bubble film by the surface of the glass! (Image, video, and research credit: P. Bourrianne et al., see also)

Snowing in the Core
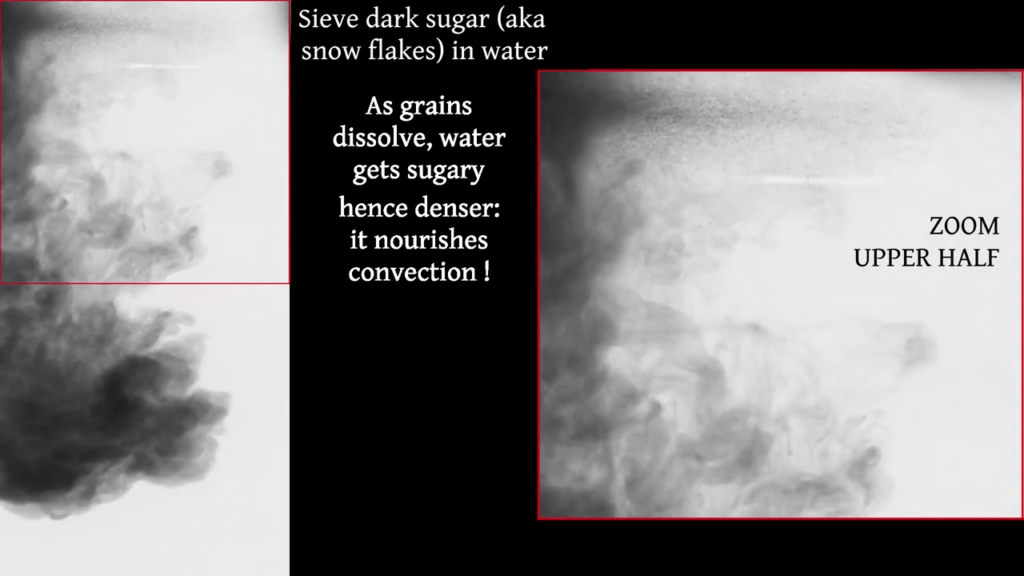
Some rocky planetary bodies, like Jupiter‘s moon Ganymede, generate magnetic fields through snow-like, solid precipitation that falls in their liquid metal cores. To study this peculiar and complex arrangement, researchers look at sugar grains falling through — and dissolving into — water. The solid sugar grains mimic the iron snowflakes that fall in Ganymede’s core. As they sink, they drag fluid with them. But the grains can also dissolve, making the fluid around them denser and prone to sinking even faster. The dense, sinking flows trigger buoyant convection inside the surrounding fluid.
As seen in the experiments, there are many factors competing here. Large grains dissolve more slowly and are able to drag more fluid with them as they fall. Small grains, on the other hand, dissolve quickly, causing more buoyancy-driven flows. Laboratory analogs like these help scientists unravel the complexities of situations we cannot observe otherwise. (Image and video credit: Q. Kriaa et al.)

Squeeze or Splatter?
Many a white shirt has met the disaster of a nearly-empty condiment bottle. One moment, you’re carefully squeezing out ketchup, and the next — sppplltlttt — you’re covered in red splatters. This messy phenomenon of gas displacing a liquid is widespread, showing up in condiments, some volcanic eruptions, and even the reinflation of a collapsed lung. Researchers have now constructed a mathematical model to fully capture and explain the process.
When you squeeze a container with both air and a liquid — like ketchup — in it, the air is easily compressed but the liquid is not. The extra pressure of the air creates a driving force that pushes the liquid out, despite its viscous resistance. Most of the time, these two forces are balanced, and the ketchup flows smoothly out of the container. But when the volume of ketchup is small compared to the air, squeezing can overpressurize the air, driving the ketchup out in an uncontrolled burst.
Luckily, the mathematics also suggest a solution to this problem: squeeze more slowly and double the size of the nozzle. You can also, they note, simply remove the top to avoid splatter. (Image credit: Rodnae Productions; research credit: C. Cuttle and C. MacMinn; via Ars Technica; submitted by Kam-Yung Soh)

Self-Propelled Droplets
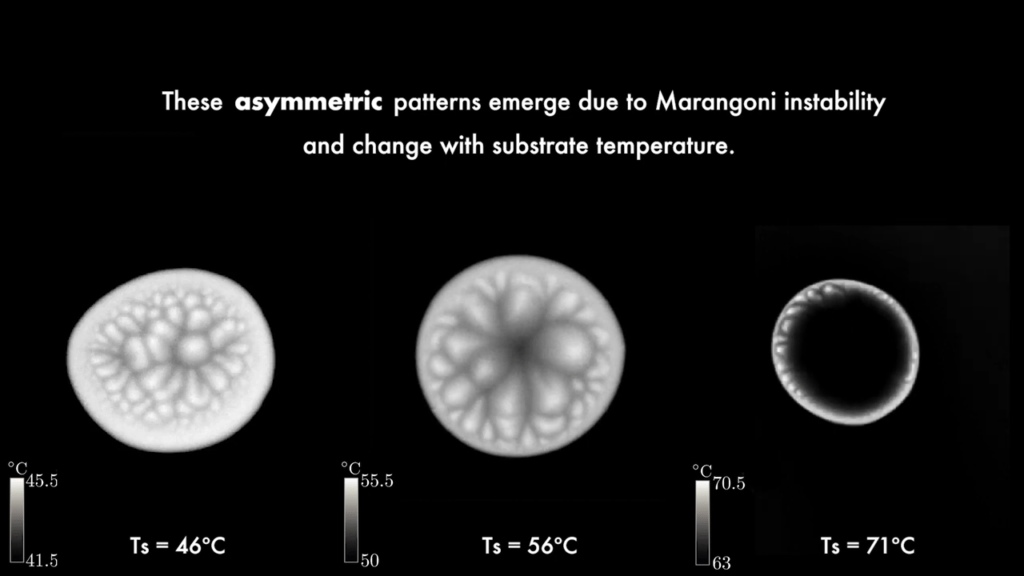
Drops of ethanol on a heated surface contract and self-propel as they evaporate. My first thought upon seeing this was of Leidenfrost drops, but the surface is not nearly hot enough for that effect. Instead, it’s significantly below ethanol’s boiling point. Looking at the drops in infrared reveals beautiful, shifting patterns of convection cells on the drop. The patterns are driven by the temperature difference along the drop; at the bottom, the drop is warmest, and at its apex, it is coldest. Those differences in temperature create differences in surface tension, which drives a surface flow that breaks the drop’s symmetry. The asymmetry, the authors suggest, is responsible for the drop’s propulsion. (Image and video credit: N. Kim et al.)

“A Sense of Scale – Reminiscence”
In so much of fluid dynamics, size does not matter. We see the same patterns mirrored across nature from a fuel injection nozzle to galactic clusters. And no one plays with that sense of scale better than artist Roman De Giuli, whose microscale practical effects give the impression of flying above glittering alien coastlines. Ink and paint squeeze around craggy islands, leaving perfect streamlines to mark their passage. Fractal fingers expand like river deltas seeking the path to the sea. Enjoy more of De Giuli’s work on his website and Instagram. (Image and video credit: R. De Giuli; via Colossal)

Nanoconfined Water
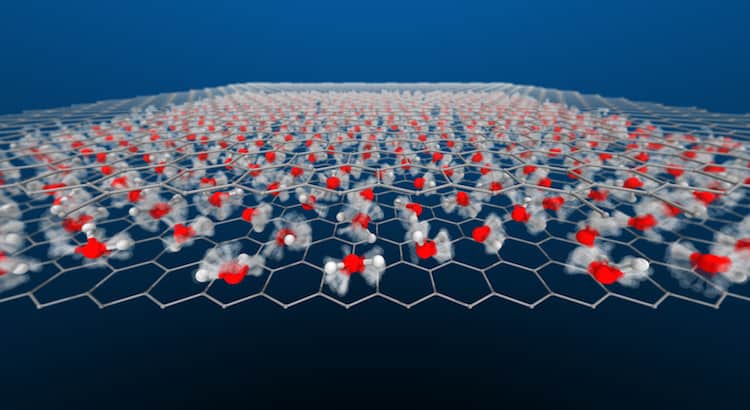
Water is a decidedly weird substance. It’s densest above its freezing point; it has a slippery liquid-like layer on its solid form; and, in the right form, it can bend like a wire. So it’s not surprising that water demonstrates some odd behaviors when it’s confined inside a space so narrow it’s only one molecule thick.
A new, simulation-based study finds that this nanoscale-confined water flows with a wide variety of behaviors, depending on the temperature and pressure. In some conditions, the water ceases to act molecularly, with hydrogen atoms flowing through a lattice of oxygen atoms. These superionic forms were thought only to exist in the extreme conditions of a gas giant’s interior, but these simulations suggest we can find them under far milder circumstances. (Image and research credit: V. Kapil et al.; via Physics World; submitted by Kam-Yung Soh)

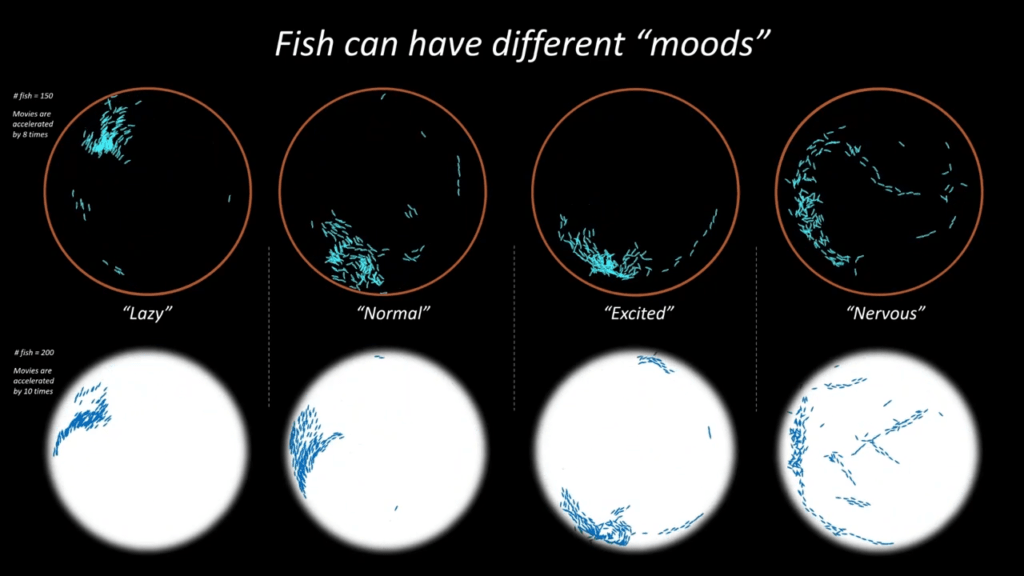
Simulating Schools
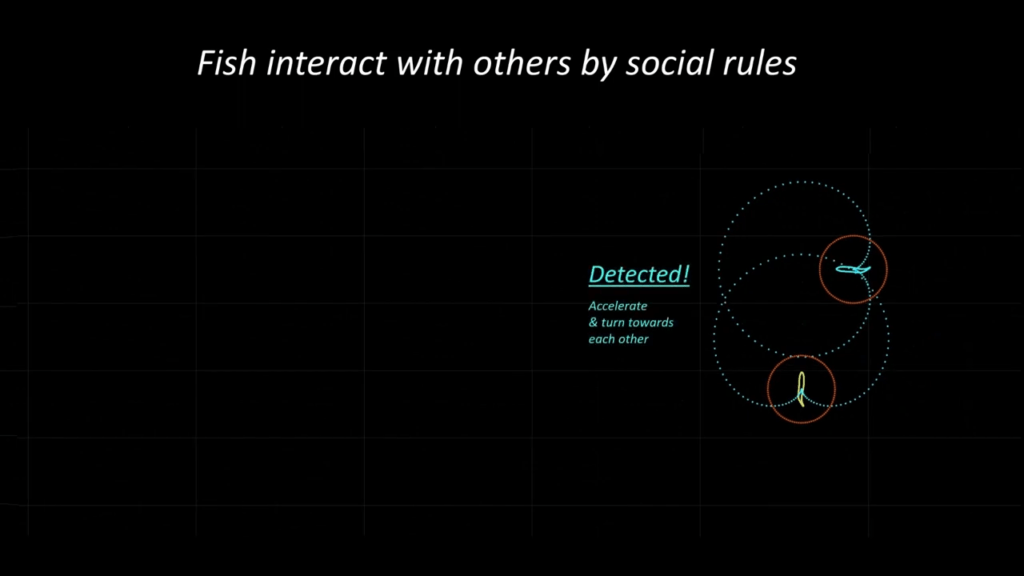
In nature, fish school for many reasons: protection from predators, increased sensing, and hydrodynamic advantages. To capture this complex behavior, researchers are building their own digital fish, governed by known rules. Here, scientists give each fish social rules — based on vision range and preferred distance from a neighbor — and hydrodynamic rules — based on a fish’s wake. With the rules in place, they can then observe the schooling behaviors of their digital fish. Like their real counterparts, these schools show different flocking based on apparent “moods”. (Image and video credit: J. Zhou et al.)

Soapy Solutions
When a drop of soap falls into a pool of water, its surface-loving molecules spread out on the water’s surface. Exactly how the soap spreads depends on the local concentration of its surfactant molecules, which create areas with different surface tensions that cause flow. All in all, it’s a tough process to predict because it varies in time at every point on the pool. But a recent paper offers a new class of exact solutions for the problem.
The paper considers a surfactant-laden droplet spreading over a (relatively speaking) deep pool. Other researchers showed recently that this situation can be described with a complex version of the Burgers’s equation, which was originally developed to describe turbulent flows. The authors solved the equation for a variety of initial conditions and found that the time-dependent spread of the surfactants was sensitive to the initial surface distribution. The higher the initial surface concentration, the faster the surfactants spread. (Image credit: T. Despeyroux; research credit: T. Bickel and F. Detcheverry; via APS Physics; submitted by Kam-Yung Soh)

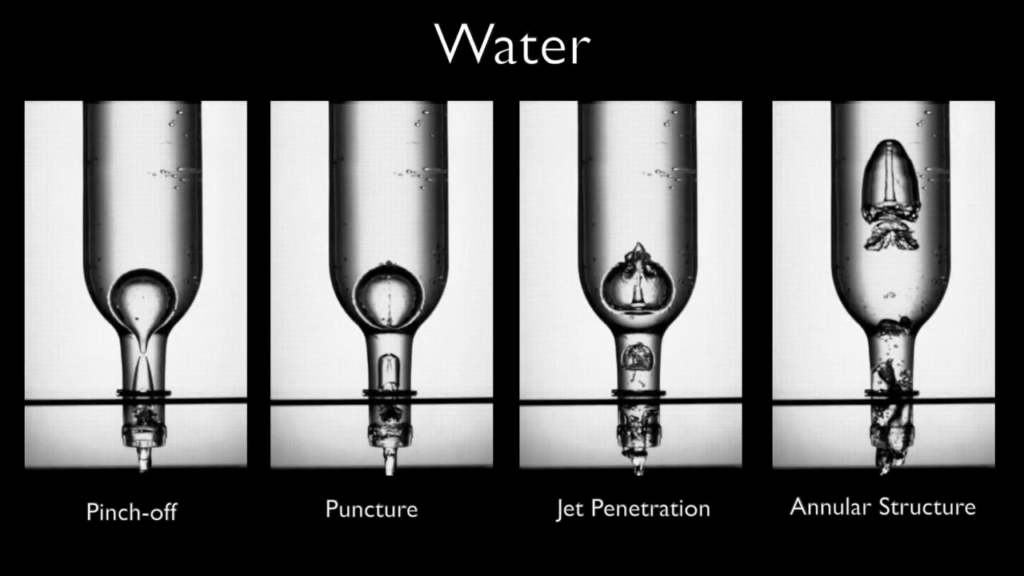
Draining a Bottle
Turn a bottle upside-down to empty it, and you’ll hear a loud glug-glug-glug as the liquid in the bottle empties and air rushes in. In this video, researchers aim a high-speed camera at the very first bubble that forms during the process. Once the bubble reaches the wider area of the bottle, it tends to pinch off in the bottle’s neck. That creates a narrow jet that pierces the bubble and flies all the way to the other side, leaving a column of liquid inside the rising bubble. Increasing the fluid’s viscosity has remarkably little effect, at least until the liquid is extremely viscous. (Image and video credit: H. Mayer et al.)